Definition
Gauge and barometric Pressure
Gauge pressure is the amount by which the pressure measured in a fluid exceeds that of the atmospheric pressure.
where is absolute pressure or gauge pressure and is atmospheric pressure.
Barometric pressure (also known as atmospheric pressure) is the force exerted by the atmosphere at a given point. It is known as the "weight of the air". A barometer measures barometric pressure. Measurement of barometric pressure can be expressed in millibars(mbar) or in inches or millimeters of mercury ().
Barometric pressure is measured by the height of mercury column.
where is absolute pressure or gauge pressure and is atmospheric pressure.
Barometric pressure (also known as atmospheric pressure) is the force exerted by the atmosphere at a given point. It is known as the "weight of the air". A barometer measures barometric pressure. Measurement of barometric pressure can be expressed in millibars(mbar) or in inches or millimeters of mercury ().
Barometric pressure is measured by the height of mercury column.
Example
Principle of Manometer and Example
A manometer is an instrument that uses a column of liquid to measure pressure
Example: The reading of a manometer fitted to a closed tap is 3.5 x 10
Nm. If the valve is opened the reading of the manometer falls to
3 x 10 Nm. What is the velocity of water?
Solution:
Applying bernouillis theorem to the fluid before and after the volume is opened,
Example: The reading of a manometer fitted to a closed tap is 3.5 x 10
Nm. If the valve is opened the reading of the manometer falls to
3 x 10 Nm. What is the velocity of water?
Solution:
Applying bernouillis theorem to the fluid before and after the volume is opened,
Definition
Units of density
Some common units of density are listed below:
- SI unit:
- CGS unit: .
Definition
Relative Density definition
Relative density is defined as the ratio of density of a substance to the density of the standard substance. Usually water at is used as a standard for a liquid or solid and air for a gas. It is a unitless quantity.
Definition
Relative Density
Density of water at is given by
Relative density,
(in CGS units)
Relative density,
(in CGS units)
Definition
Uses of relative density
Following are the uses of relative density:
1) It is used for determining the density of unknown substance from the known density of another substance.
2) It is used by geologist to find out the mineral content into the rock.
3) Testing purity of a substance (example gold).
1) It is used for determining the density of unknown substance from the known density of another substance.
2) It is used by geologist to find out the mineral content into the rock.
3) Testing purity of a substance (example gold).
Definition
Define and explain hydrostatic paradox
The barometric formula depends only on the height of the fluid chamber, and not on its width or length. Given a large enough height, any pressure may be attained. This feature of hydrostatics has been called the hydrostatic paradox. As expressed by W. H. Besant, "Any quantity of liquid, however small, may be made to support any weight, however large."
The hydrostatic paradox is explained by the fact that, since the hydrostatic pressure is always normal to a vessels walls, the force of the pressure on the inclined walls has a vertical component that compensates for the excess weight of the volume of fluid in a vessel that is wider at the top than a cylindrical vessel with the same bottom area and compensates for the lack of weight of the volume of fluid in a vessel that is narrower at the top.
The hydrostatic paradox is explained by the fact that, since the hydrostatic pressure is always normal to a vessels walls, the force of the pressure on the inclined walls has a vertical component that compensates for the excess weight of the volume of fluid in a vessel that is wider at the top than a cylindrical vessel with the same bottom area and compensates for the lack of weight of the volume of fluid in a vessel that is narrower at the top.
Result
Hydraulic press
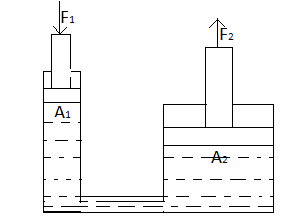
The hydraulic press works on the principle of Pascal's law. The pressure throughout a closed system is constant since there is no leakage. One part of the system is a piston acting as a pump, with a modest mechanical force acting on a small cross-sectional area, the other part is a piston with a larger area which generates a correspondingly large mechanical force. The larger force is used to press cotton or any other similar material against a fixed roof.
Result
Hydraulic jack

The hydraulic jack works on Pascal's law. An effort is applied on the piston of the jack with smaller area. This results in a greater force on the other end of the jack and hence results in a multiplication of force.
In the given diagram,
Since,
In the given diagram,
Since,
Result
Hydraulic brake

The hydraulic brake works on the principle of transmission of pressure by Pascal's Law. When the brake is pressed by applying a force the pressure in the braking fluid is increased. This is transmitted to the pistons with larger area which press against the break shoes () and the friction between the shoes and the rotor causes a braking torque to be generated, slowing the vehicle.
| BookMarks |
Page 11 Page 12 Page 13 Page 14 Page 15 Page 16 Page 17 Page 18
0 Comments
Post a Comment